
Source: To find formal charge, take the valence electrons of the atom, and subtract these things from it: Calculate the formal charge of the compound using the lewis dot structure in step 1 and the formula given. Source: Using the formula charge formula for each atom present, we can calculate. Source: The given formula can calculate the formal charge on each atom: Formal charge is the charge of an atom in a molecule. Source: Formal charge is the charge of an atom in a molecule. 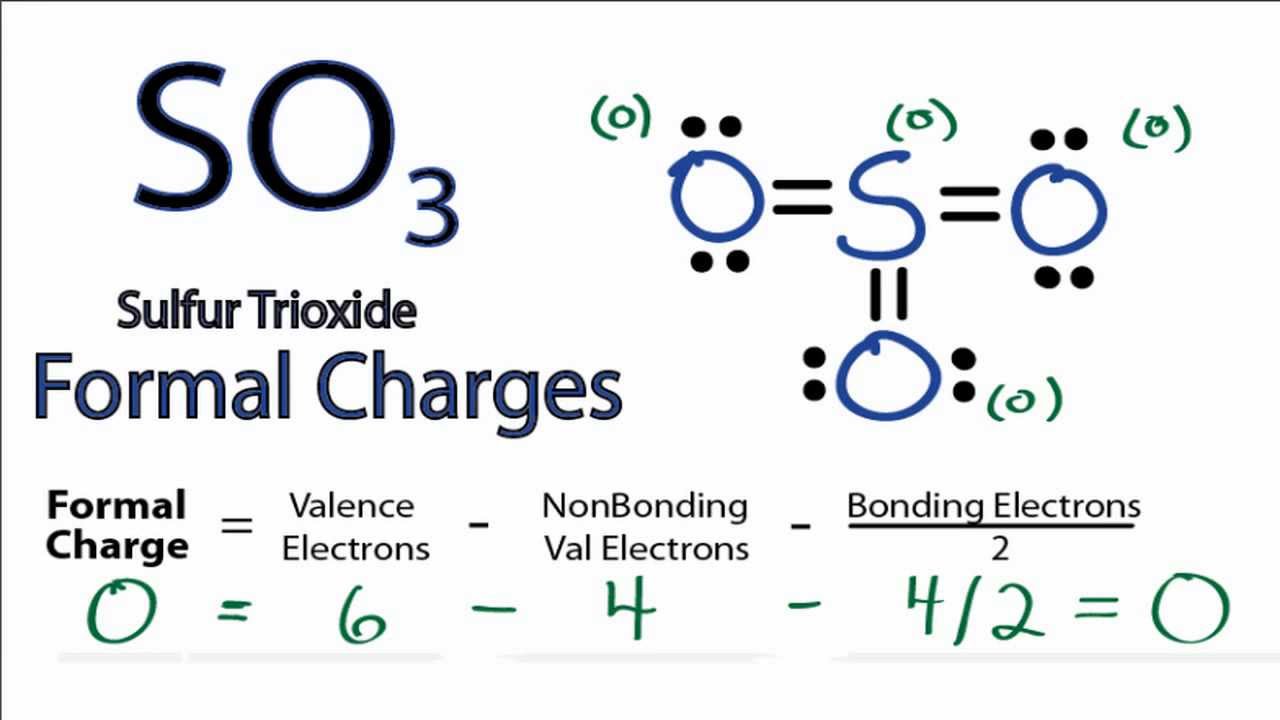
For example, in nh 3, n has 1 lone pair (2 electrons) and 3. Source: Reading about how to draw lewis structures, all i know is that formal charge is the charge assigned to an atom in a molecule assuming that electrons are shared equally. Source: Īn atom can have the following charges: The positive or negative or neutral (zero) charges, present on the atoms of any lewis structure or molecule is known as the formal charge. Source: The formal charges present in each of these molecular structures can help us pick the most likely arrangement of atoms. To find formal charge, take the valence electrons of the atom, and subtract these things from it: When that happens, we usually assign formal charges to the bonded atoms to help determine the correct lewis structure. Calculating NO3 Formal Charges Calculating Formal Charges for NO3 from Divide the electron pairs in bonds equally for all the bonds. Possible lewis structures and the formal charges for each of the three. How To Find Formal Charge In Lewis Structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
